NAPE-Phospholipase D inhibitor (LEI-401) small molecule (tool compound)
Invented by Mario Van Der Stelt
Invented at Leiden University and Leiden University Medical Center
- Datasheet
- References (1)
- Inventor Info
Info
| Catalogue Number | 160829 |
| Antigen/Gene or Protein Targets | N-acyl phosphatidylethanolamine-specific phospholipase D |
| Synonyms | N-acyl phosphatidylethanolamine-specific phospholipase D inhibitor, LEI-401 |
| Type | Inhibitor |
| Relevance |
D-type phospholipase that hydrolyzes N-acyl-phosphatidylethanolamines (NAPEs) to produce bioactive N-acylethanolamines/fatty acid ethanolamides (NAEs/FAEs) and phosphatidic acid. Cleaves the terminal phosphodiester bond of diacyl- and alkenylacyl-NAPEs, primarily playing a role in the generation of long-chain saturated and monounsaturated NAEs in the brain._x000D_ May control NAPE homeostasis in dopaminergic neuron membranes and regulate neuron survival, partly through RAC1 activation. As a regulator of lipid metabolism in the adipose tissue, mediates the crosstalk between adipocytes, gut microbiota and immune cells to control body temperature and weight. In particular, regulates energy homeostasis by promoting cold-induced brown or beige adipocyte differentiation program to generate heat from fatty acids and glucose. Has limited D-type phospholipase activity toward N-acyl lyso-NAPEs |
| On Target IC50 | 27 nM |
| Selectivity | IC50> 10uM for CB1R & CB2R, FAAH, PLA2G4E, DAGL, MAGL, ABHD6 |
| Molecular Formula | C24H31N5O2 |
| Key Attributes | Reduces N-acylethanolamines levels, including anandamide, in cells and mouse brain |
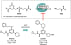
| lUPAC | N-(Cyclopropylmethyl)-6-((S)-3-hydroxypyrrolidin-1-yl)-2-((S)-3-phenylpiperidin-1-yl)pyrimidine-4-carboxamide. |
| Molecular Weight (g/mol) | 422 |
| In vivo applications | good bioavailability F (i.p.) = 48 % and brain penetrant |
| In vitro applications | good cellular penetration |
| Solubility | 1.7 mg/L |
| Research Area | Neurobiology, Metabolism |
| Notes | Mock et al., Nature Chemical Biology, 2020, 16, 667-675 |
References: 1 entry
References: 1 entry
Inventor Information
Inventors

|
Mario Van Der Stelt |